What packaging equipment is used for non-sterile medical devices, OTC diagnostic test kits, and hospital supply bundles? Light medical device and healthcare product packaging — particularly consumer-facing, non-sterile items distributed through pharmacy and retail channels — uses the same flow-wrapping, shrink-wrapping, and tray-sealing processes that serve food and consumer goods. Tongue depressors, bandages, gauze pads, diagnostic test kits, and OTC medical product bundles all move through standard packaging equipment, with material selections and format configurations appropriate to healthcare product requirements.
For medical devices requiring validated sterile packaging processes, operators must consult applicable FDA regulatory requirements and medical device packaging standards before selecting equipment. The applications described here are appropriate for non-sterile, consumer-facing healthcare products distributed through retail and pharmacy channels.
Healthcare Product Packaging Applications
Single-Use Medical Item Wrapping — Tongue Depressors, Bandages, Gauze Pads
Tongue depressors, bandages, gauze pads, disposable thermometer sheaths, and other single-use medical items are flow-wrapped on the Series 90 or 150 in medical-grade poly film. The sealed wrap provides dust and contamination protection for items distributed to clinics, pharmacies, and hospitals. Individual wrap formats allow single-item dispensing from bulk distribution packaging.
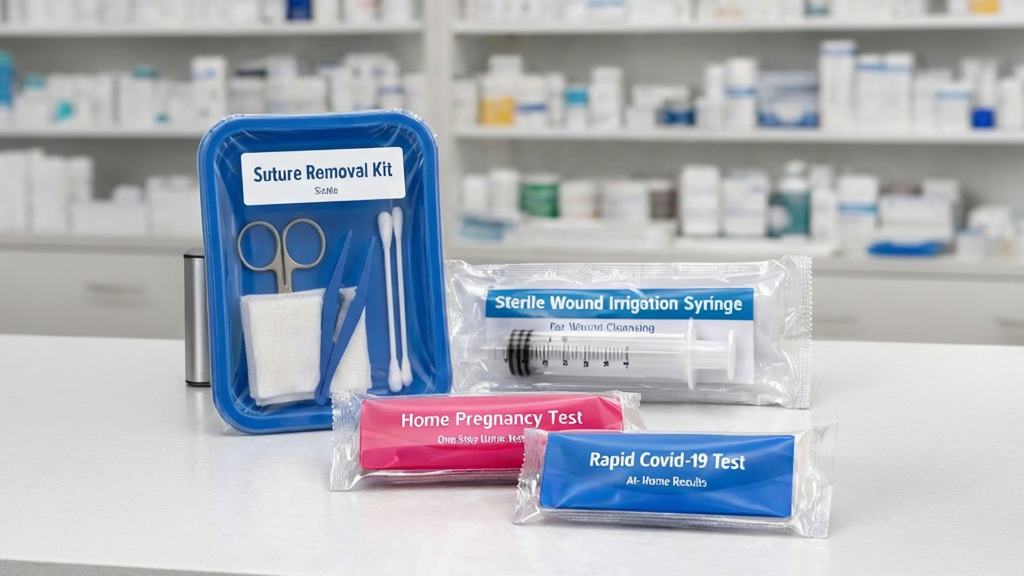
Medical Device and Diagnostic Kit Tray Packaging
Small medical devices, diagnostic test components, and procedural kits with multiple parts are sealed in APET trays on the Series 30 or 60. The sealed tray format keeps multi-component kits organized, protects contents from handling damage, and provides tamper evidence. The clear film lid provides full product visibility — important for medical kits where users need to verify contents before opening the package.
OTC Diagnostic Test Kit Overwrapping — Rapid Tests, Blood Glucose Strips
Rapid test kits, blood glucose test strip packs, and similar OTC diagnostic products are flow-wrapped or shrink-wrapped for retail pharmacy distribution. The wrapped format adds tamper evidence — a practical and sometimes regulatory requirement for OTC diagnostic products — and provides branding real estate on the outer wrap for product identification and instructions.
Multi-Unit Hospital Supply Bundle Shrink Wrapping
Disposable glove packs, syringe bundles, and other multi-unit hospital supply items are shrink-wrapped on the Series 150 for distribution to healthcare facilities. The shrink wrap consolidates units, reduces secondary packaging material, and maintains bundle integrity through the distribution chain from manufacturer to hospital supply room.
Which AmeriPak Equipment Is Right for Healthcare Products?
Series 90 / Series 150 Horizontal Flow Wrapper — Single-Use Items and Diagnostic Test Kits
Single-use medical item wrapping, OTC diagnostic kit overwrapping. Compatible with medical-grade poly films and a wide range of product dimensions.
Series 30 / Series 60 Tray Sealer — Multi-Component Medical Kits and Diagnostic Products
Multi-component medical kit tray sealing with clear lidding film for component visibility and tamper evidence.
Series 150 Shrink Film Wrapper — Hospital Supply Bundles and OTC Kit Bundling
Multi-unit hospital supply bundles, OTC product kit bundling, retail-ready healthcare product shrink wrapping.
Important Note on Medical Device Packaging Regulations
For medical devices subject to FDA device regulations, packaging validation requirements under ISO 11607 and related standards govern the selection and validation of packaging equipment and materials. AmeriPak equipment is not validated for sterile barrier packaging applications. Operators packaging regulated medical devices must consult their quality and regulatory teams before specifying packaging equipment and processes.
The applications on this page are appropriate for non-sterile, consumer-facing healthcare products — OTC diagnostics, first aid supplies, hospital consumables — distributed through retail and healthcare facility channels
Frequently Asked Questions — Medical Device and Healthcare Packaging Equipment
Is AmeriPak equipment appropriate for packaging sterile medical devices?
AmeriPak equipment is designed and marketed for food and general consumer goods packaging. For sterile medical device packaging applications governed by ISO 11607 and FDA device regulations, specialized validated sterile barrier packaging equipment and processes are required. Consult your regulatory and quality teams for sterile device packaging requirements. AmeriPak equipment is appropriate for non-sterile healthcare products and consumer-facing OTC items.
We package OTC diagnostic test kits for retail pharmacy. Is this a suitable application for AmeriPak equipment?
Yes. OTC diagnostic test kits — rapid tests, blood glucose strips, pregnancy tests — packaged for retail pharmacy distribution are a straightforward application for AmeriPak flow wrappers and tray sealers. The packaging process is similar to consumer goods: overwrapping or tray sealing non-sterile components for retail distribution. Confirm tamper-evidence and labeling requirements with your regulatory counsel.
Ready to solve your packaging challenge? Use our Contact Page or Call AmeriPak at 215-792-7272 – Tray sealers, flow wrappers, and shrink wrappers — made in the USA in Warminster, PA.
